The landscape of antibody discovery is rapidly evolving, moving away from slow, iterative binding-focused methods toward a paradigm of function-first, high-throughput assessment. Two technologies are driving this transition: advances in artificial intelligence (AI) for antibody design, and high-throughput, single‑cell functional screening platforms such as the Beacon® system. Together, they are redefining how we explore the vast antibody fitness landscape. The result is faster decisions, better leads, and stronger data to guide the next round of AI designs.
The Parallel Revolution: AI Design and Optofluidic Functional Assays
Traditional methods like phage display often prioritize discovering binders before attempting functional characterization and/or antibody engineering using limited toolsets reliant on past expertise. These binder first workflows take weeks to months producing and testing many sequences that bind recombinant targets but later fail on function, stability, or specificity, adding cost and delays. Teams need a way to predict and verify function earlier, with clearer signals before heavy investment. The integration of AI and advanced antibody characterization allows for simultaneous optimization of function and developability.
- The AI Impact: Generative models are now used to design novel antibody sequences from scratch, focusing on unexplored regions of the antibody space. These models don’t just find binders; they can predict structural integrity and binding orientations in seconds1. In addition, AI tools can be used to iterate on antibody design, taking advantage of more sophisticated antibody characterization tools to engineer antibodies with reduced immunogenicity and liabilities, and more desirable binding affinity, stability, and solubility.
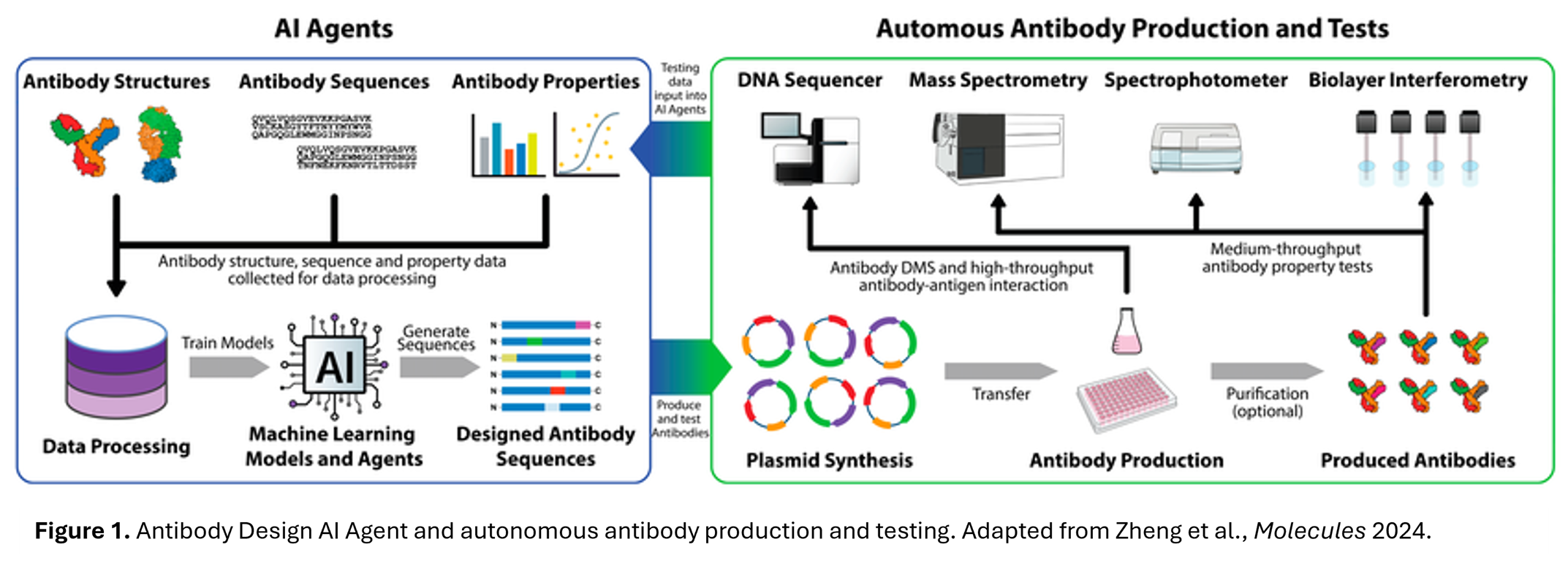
- The Beacon Impact: In parallel, the Beacon optofluidic system has revolutionized the discovery of novel antibody candidates. By utilizing opto-electrical positioning (OEP) to move single B cells into tens of thousands of nanoliter-sized NanoPen chambers, researchers can perform both binding and functional assays on-chip. The Beacon system screens tens of thousands of individual cells simultaneously, assessing antigen specificity, cross-reactivity, relative affinity, and secretion rates within hours, as well as functional attributes like internalization and ligand blocking.
Keeping Pace with AI Discoveries with High-Throughput Functional Assessment
Machine learning models now include “filtering mechanisms” that score and select candidates based on solubility, thermostability, and aggregation propensity before they ever reach a lab bench. The Beacon system mirrors this “early-stage validation” by allowing researchers to see a cell’s functional phenotype (how it behaves) before committing to sequencing. This ensures that only the most therapeutically relevant clones (i.e., those with the desired functional activity) are moved forward.
The ultimate efficiency is found in workflows pairing the two. In this model, AI-designed candidates are expressed and evaluated on a medium-throughput scale (500–1,000 candidates), testing for binding affinity and poly-reactivity. The high-quality functional data produced by systems like the Beacon system can serve as the “ground truth” to train these AI agents, refining future generations of digital designs. In addition to discovering functional antibodies from primary B cells, the Beacon system can accelerate the AI design-build-test loop through functional analysis of mammalian libraries of AI-designed functional antibodies.
Spotlight: Identifying Cross Reactive Antibodies
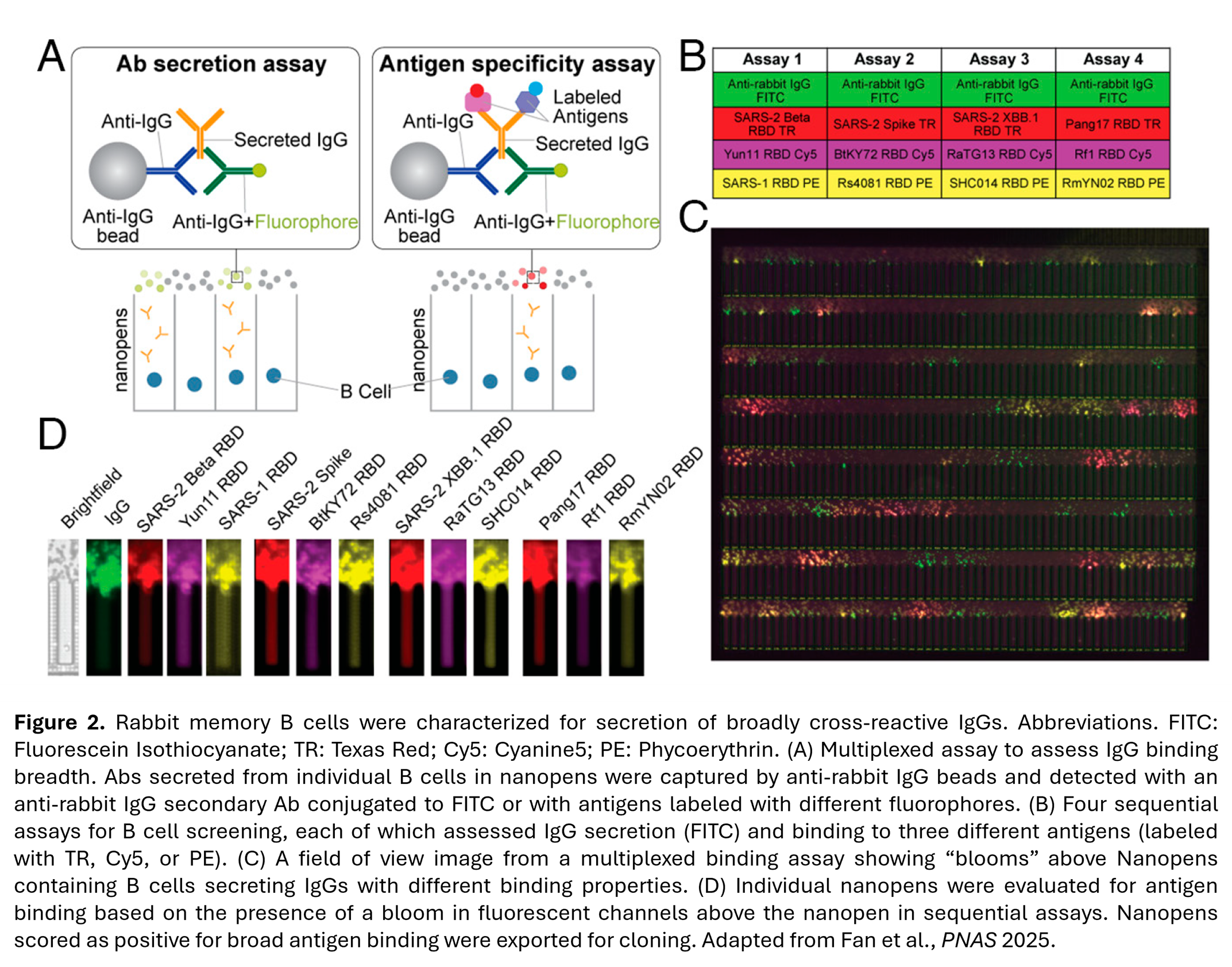
In a recent publication, researchers at the California Institute of Technology used the Beacon system to showcase the impact of pairing of high-throughput single-cell screening and functional assays to identify cross-reactive monoclonal antibodies2 (Figure 2). Researchers screened approximately 27,000 activated primary memory B cells and multiplexed assays to profile binding to 12 antigens by IgGs secreted by individual B cells. This screening aimed to determine breadth of responses following two immunization strategies to “focus” the immune response on conserved regions to identify the “best-of-the-best” cross-reactive candidates. This “functional-first” approach allowed them to identify and export B cells based on their breadth of binding. For instance, they could specifically target rare B cells that recognized up to 11 different RBDs. This is a significant shift from traditional methods that require cloning and expressing thousands of antibodies before testing their breadth.
A New Benchmark for Efficiency
The combined impact of these technologies is enhancing antibody discovery and shortening the costs and risks to therapeutic development and research. Where traditional workflows might take six to nine months, an integrated approach using AI-driven design and optofluidic validation can reduce timelines, often yielding high-quality, developable candidates in just a few weeks. We are no longer spending years searching for a sequence; we can instead design the cells and use light-driven precision to verify functionality. By making high-throughput, cell-based screening the new standard, the industry is ensuring that the next generation of biologics is not only discovered faster and cost effective but is therapeutically more effective for the patients who need them.
References
-
- [1] Zheng, J., Wang, Y., Liang, Q., Cui, L., & Wang, L. (2024). The Application of Machine Learning on Antibody Discovery and Optimization. Molecules, 29(24), 5923. https://doi.org/10.3390/molecules29245923.
- [2] Fan, C., Keeffe, J. R., Malecek, K. E., Cohen, A. A., West, A. P., Jr, Baharani, V. A., Rorick, A. V., Gao, H., Gnanapragasam, P. N. P., Rho, S., Alvarez, J., Segovia, L. N., Hatziioannou, T., Bieniasz, P. D., & Bjorkman, P. J. (2025). Cross-reactive sarbecovirus antibodies induced by mosaic RBD nanoparticles. Proceedings of the National Academy of Sciences of the United States of America, 122(21), e2501637122. https://doi.org/10.1073/pnas.2501637122.


